Inside Triethanolamine And Its Application
Triethanolamine, often abbreviated as TEA, is aviscous organic compound that is both a tertiaryamine and a triol. A triol is a molecule with three alcohol groups.
It is a colourless compound although samples may appear yellow because of impurities.
Production Triethanolamine
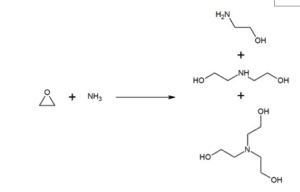
triethanolamine production
Triethanolamine is produced from the reaction of ethylene oxide with aqueous ammonia, also produced are ethanolamine and diethanolamine. The ratio of the products can be controlled by changing thestoichiometry of the reactants.
Applications
Triethanolamine is used primarily as an emulsifier and surfactant. It is a common ingredient in formulations
Triethanolamine is used primarily as an emulsifier and surfactant. It is a common ingredient in formulations
used for both industrial and consumer products. The triethanolamine neutralizes fatty acids, adjusts and buffers the pH, and solubilises oils and other ingredients that are not completely soluble in water. Some common products in which triethanolamine is found are liquid laundry detergents, dishwashing liquids, general cleaners, hand cleaners, polishes, metalworking fluids, paints, shaving cream and printing inks.
Cement production
Triethanolamine is also used as organic additive (0.1 wt. %) in the grinding of cement clinker. It facilitates
the grinding process by preventing agglomeration and coating of the powder at the surface of balls and mill wall.
Triethanolamine is also used as organic additive (0.1 wt. %) in the grinding of cement clinker. It facilitates
the grinding process by preventing agglomeration and coating of the powder at the surface of balls and mill wall.
Cosmetics and medicine
Various ear diseases and infections are treated with eardrops containing triethanolamine polypeptide oleatecondensate,
such as Cerumenex in the United States. In pharmaceutics, triethanolamine is the
active ingredient of some ear drops used to treat impactedearwax. It also serves as a pH balancer in many different cosmetic products ranging from cleansing creams and milks, skin lotions, eye gels, moisturizers, shampoos, shaving foams etc. TEA is a fairly strong base: a 1% solution has a pH of approximately 10, whereas the pH of skin is below pH 7, more or less 5.56.0.
Cleansing milk/cream emulsions based on TEA are particularly good at removing makeup.
Various ear diseases and infections are treated with eardrops containing triethanolamine polypeptide oleatecondensate,
such as Cerumenex in the United States. In pharmaceutics, triethanolamine is the
active ingredient of some ear drops used to treat impactedearwax. It also serves as a pH balancer in many different cosmetic products ranging from cleansing creams and milks, skin lotions, eye gels, moisturizers, shampoos, shaving foams etc. TEA is a fairly strong base: a 1% solution has a pH of approximately 10, whereas the pH of skin is below pH 7, more or less 5.56.0.
Cleansing milk/cream emulsions based on TEA are particularly good at removing makeup.
In the laboratory and in amateur photography
Another common use of TEA is as a complexing agent for aluminium ions in aqueous solutions. This reaction is often used to mask such ions beforecomplexometric titrations with another chelating agent such as EDTA. TEA has also been used in photographic (silver halide) processing. It has been promoted as a useful alkali by amateur photographers.
In Holography.
TEA is used to provide a sensitivity boost to silver halide based holograms. Also as a swelling agent to colour shift holograms. You can get the sensitivity boost without the color shift by rinsing out the TEA before
squeegee and drying. In electroless plating
squeegee and drying. In electroless plating
TEA is now commonly and very effectively used as a complexing agent in electroless plating.
Safety and regulation
Allergic reactions A 1996 study found that triethanolamine (TEA) occasionally causes contact allergy A 2001 study found
TEA in a sunscreen caused an allergic contact dermatitis.
Allergic reactions A 1996 study found that triethanolamine (TEA) occasionally causes contact allergy A 2001 study found
TEA in a sunscreen caused an allergic contact dermatitis.
A 2007 study found TEA in ear dropscaused a contact allergy.[ Systemic and respiratory tract (RT) toxicity was analyzed for 28 days in a nose specific
inhalation 2008 study in Wistar rats; TEA seems to be less potent in regard to systemic toxicity and RT
inhalation 2008 study in Wistar rats; TEA seems to be less potent in regard to systemic toxicity and RT
Irritancy than diethanolamine (DEA). Exposure to TEA resulted in focal inflammation, starting in single male animals from 20 mg/m3 concentrations.
A 2009 study stated patch test reactions reveal a slight irritant potential instead of a true allergic response in several cases and also indicated the risk of skin sensitization to TEA seems to be very low.
Tumors Reports indicated that TEA causes an increased incidence of tumor growth in the liver in female B6C3F1
mice, but not in male mice or in Fischer 344 rats A 2004 study concluded “TEA may cause liver tumors in mice via a cholinedepletion mode of action and that this effect is likely caused by the inhibition of choline uptake by cells.”
A 2009 study stated patch test reactions reveal a slight irritant potential instead of a true allergic response in several cases and also indicated the risk of skin sensitization to TEA seems to be very low.
Tumors Reports indicated that TEA causes an increased incidence of tumor growth in the liver in female B6C3F1
mice, but not in male mice or in Fischer 344 rats A 2004 study concluded “TEA may cause liver tumors in mice via a cholinedepletion mode of action and that this effect is likely caused by the inhibition of choline uptake by cells.”
Environmental toxicity
A 2009 study found that TEA has potential acute, subchronic and chronic toxicity properties in respect to aquatic species.
Regulation
TEA is listed under Schedule 3, part B of the Chemical Weapons Convention as it can be used in the
manufacture of nitrogen mustards, particularly HN3.
A 2009 study found that TEA has potential acute, subchronic and chronic toxicity properties in respect to aquatic species.
Regulation
TEA is listed under Schedule 3, part B of the Chemical Weapons Convention as it can be used in the
manufacture of nitrogen mustards, particularly HN3.
More details to come












No comments